Observation of Enhanced Chiral Asymmetries in the Inner-Shell Photoionization
of Uniaxially Oriented Methyloxirane Enantiomers
|
Most large molecules are chiral in their structure: they exist as two enantiomers, which are mirror images of each other. Whereas the rovibronic sublevels of two enantiomers are almost identical (neglecting a minuscular effect of the weak interaction), it turns out that the photoelectric effect is sensitive to the absolute configuration of the ionized enantiomer. Indeed, photoionization of randomly oriented enantiomers by left or right circularly polarized light results in a slightly different electron flux parallel or antiparallel with respect to the photon propagation direction - an effect termed photoelectron circular dichroism (PECD). Our comprehensive study demonstrates that the origin of PECD can be found in the molecular frame electron emission pattern connecting PECD to other fundamental photophysical effects such as the circular dichroism in angular distributions (CDAD). Accordingly, distinct spatial orientations of a chiral molecule enhance the PECD by a factor of about 10.
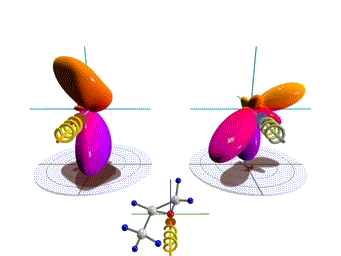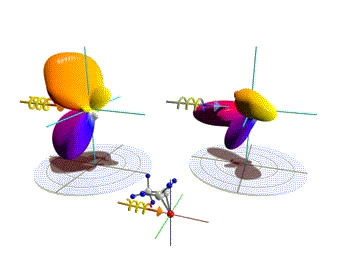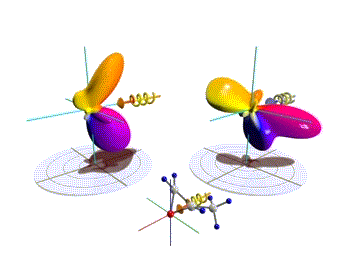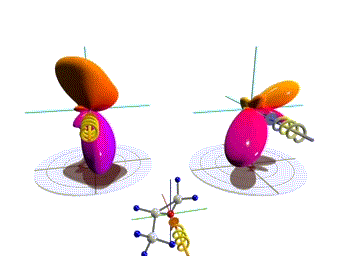
Figure 1: Three-dimensional molecular frame photoelectron angular distributions. Theoretical distributions computed here for the O 1s-ionization of R(+) methyloxirane by left (left side) and right (right side) circularly polarized light. The molecules are oriented as depicted in the insets. Observation
of Enhanced Chiral Asymmetries in the Inner-Shell Photoionization of Uniaxially Oriented Methyloxirane Enantiomers |