Direct Determination of Absolute Molecular Stereochemistry
in Gas Phase by Coulomb Explosion Imaging
|
We recently developed a method for directly imaging the handedness of simple chiral molecules. Molecules are called chiral if they appear in two structures (enantiomers) that are mirror images of each other and that cannot be brought into overlap by rotation or translation. The investigation of chirality is a vital topic in chemistry and a lot of effort is put into developing techniques to distinguish enantiomers. Up to now, however, there are few methods that allow to tell which microscopic structure (absolute configuration) is present in a sample. Especially for most liquid or gaseous substances, only indirect methods are available so far. We report a new approach that allows the determination of absolute configuration even on a single-molecule level and demonstrate its effectiveness for the prototypically chiral molecules CHBrClF and CHBr35Cl37Cl. In the focus of an intense femto-second laser pulse, the molecule under investigation is multiply ionised. This leads to complete break-up with the positively charged atomic ions repelling each other (Coulomb Explosion). The three-dimensional momentum vectors of all ions are measured in coincidence in a COLTRIMS-Setup. As can be seen by the two peaks in Figure 1, these momenta allow a clear separation between the two enantiomers. The angle θ shows if the the ions Cl+, Br+, F+ form a right-handed (R-enantiomer) or a left-handed (S-enantiomer) coordinate system. 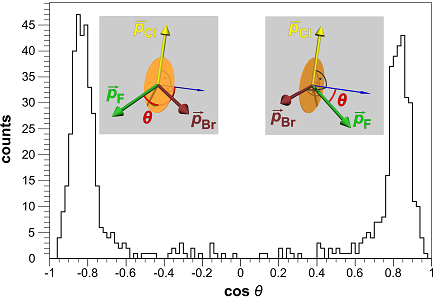 Figure 1: Chiral discrimination for CHBrClF. The angle θ is defined as whereas the one at positive values corresponds to the R enantiomer. Figure 2 shows the momenta being normalised and rotated to the molecular frame, demonstrating that the ions are mostly repelled in the direction of bond angles. The results open new perspectives for chemical and pharmaceutical diagnostics as well as for the investigation of chirality on the fundamental level.  Figure 2: Ion momenta in the molecular frame for the S-enantiomer (left) and R-enantiomer (right) of CHBrClF. The color codes are as follows: Br (red), Cl (yellow), F (green), H (white) and C (black).
Publication
Collaboration |